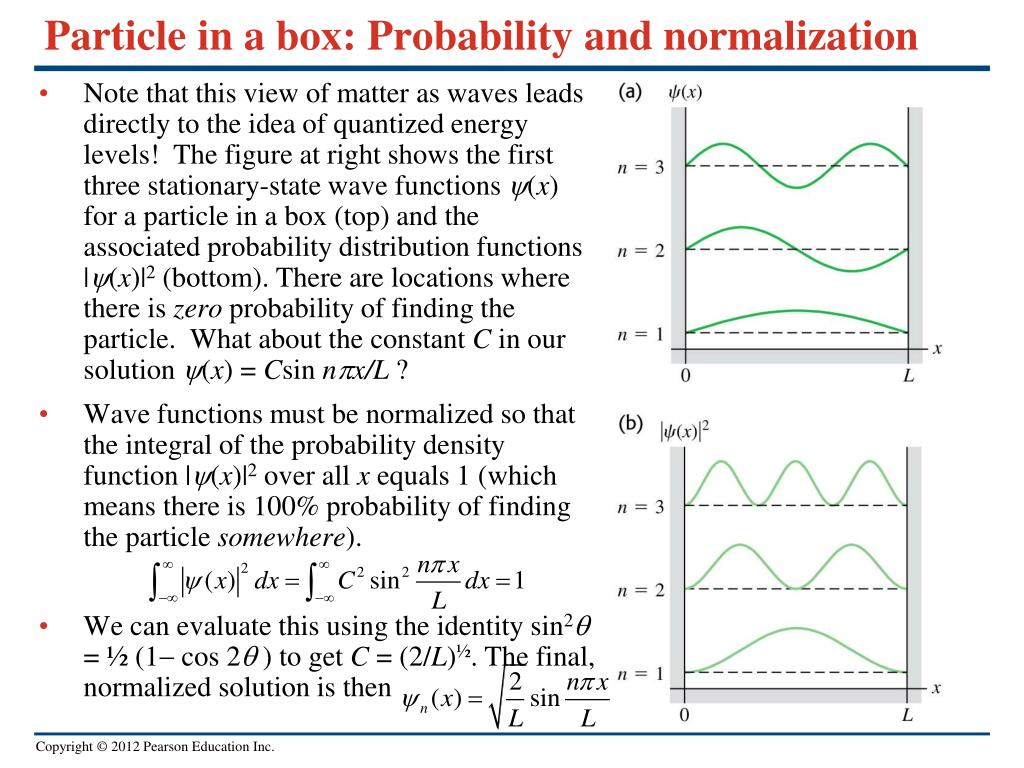
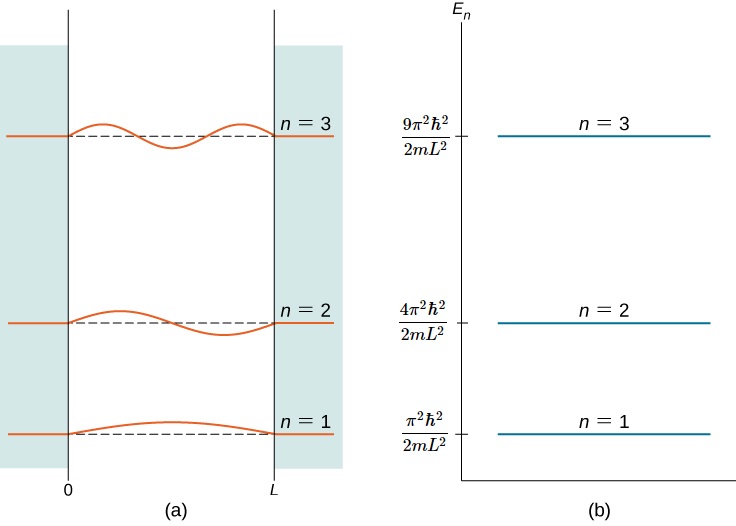
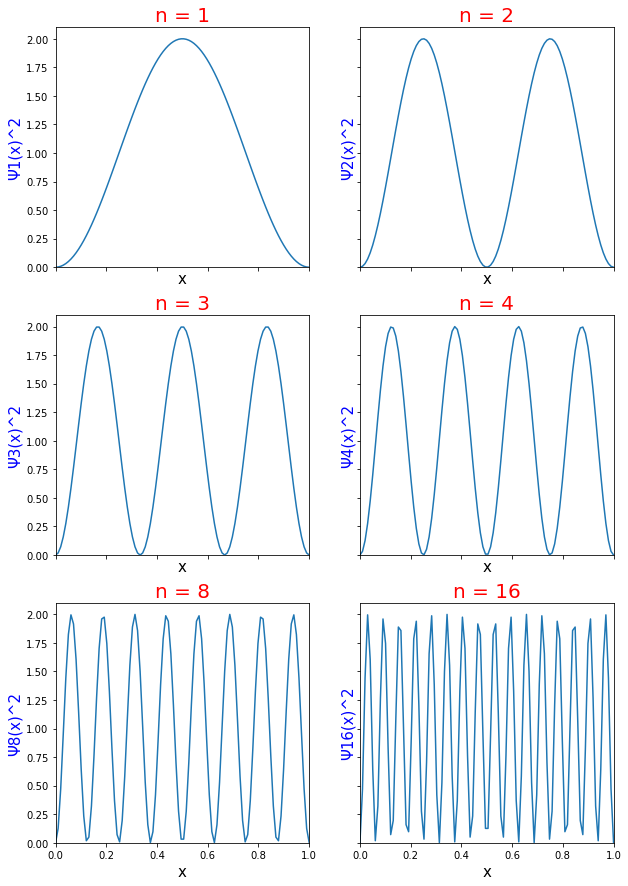
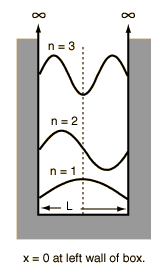
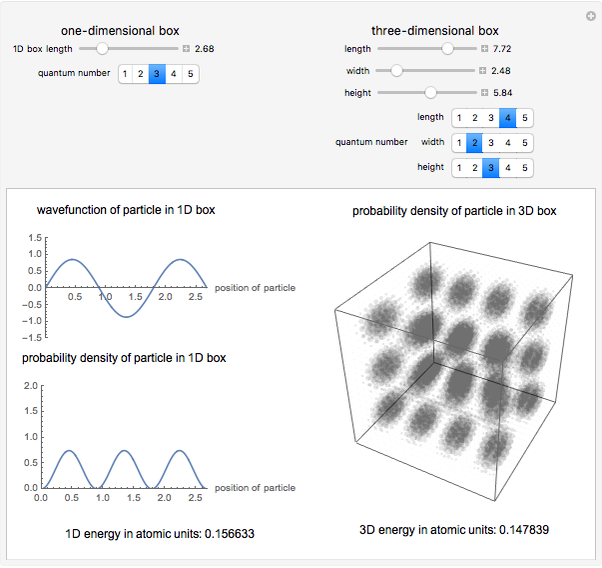
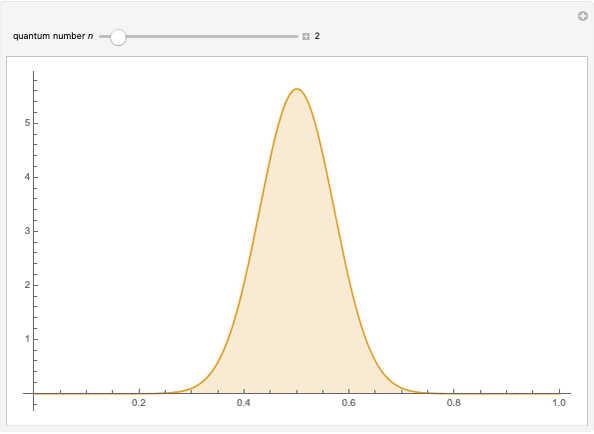
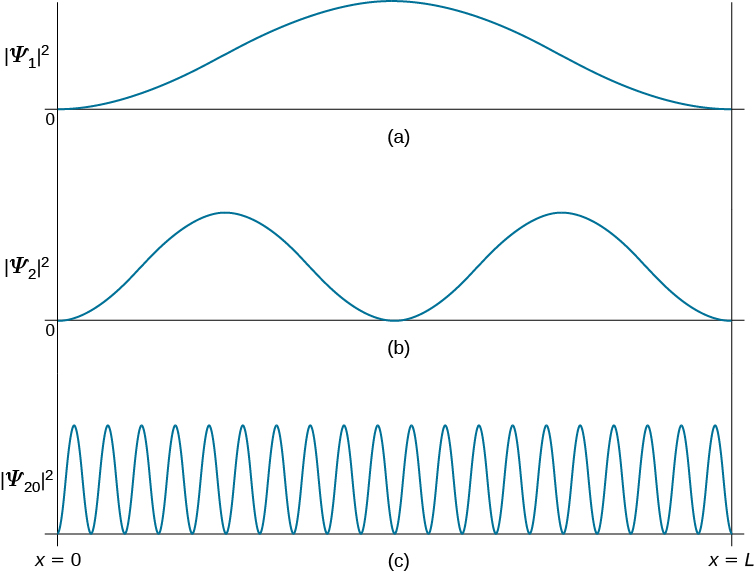
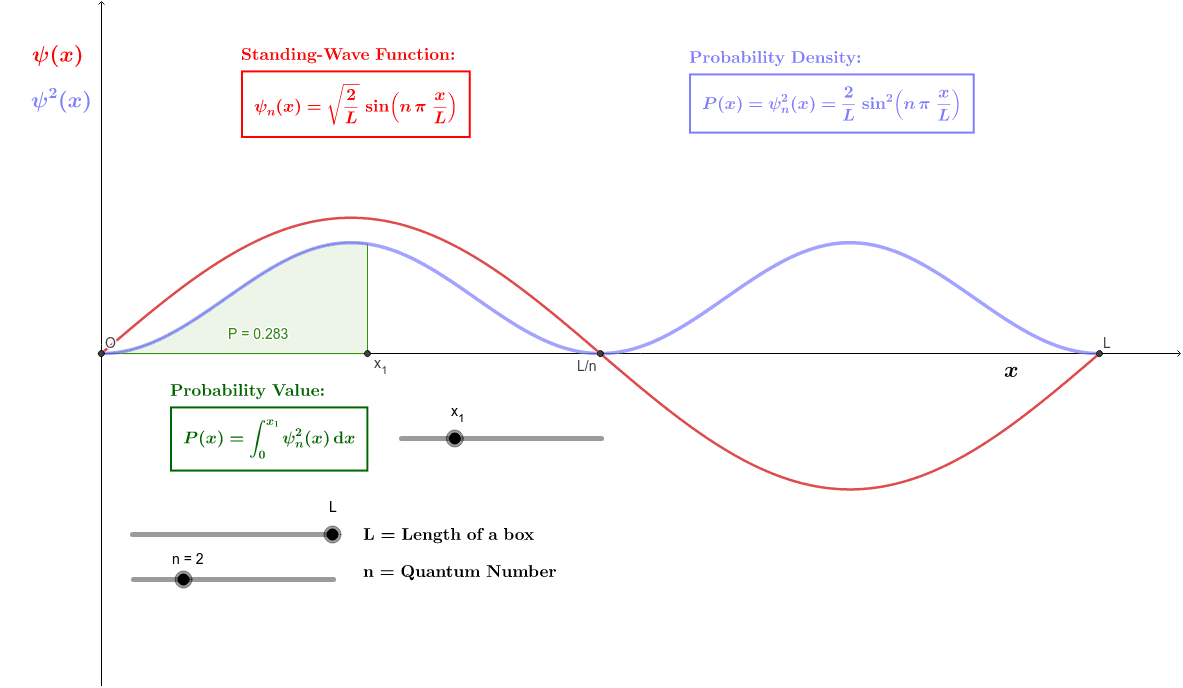
Calculate the probability of finding the particle trapped in a box of length L in the region from x=0.45L to 0.55L for the ground state and the first excited state.___ | Snapsolve
A particle is confined to a 1D box of unit length.[math] \psi(x) = \sqrt{2} [/math] till x=0.5. Find the probability of the particle having energy E3. - Quora
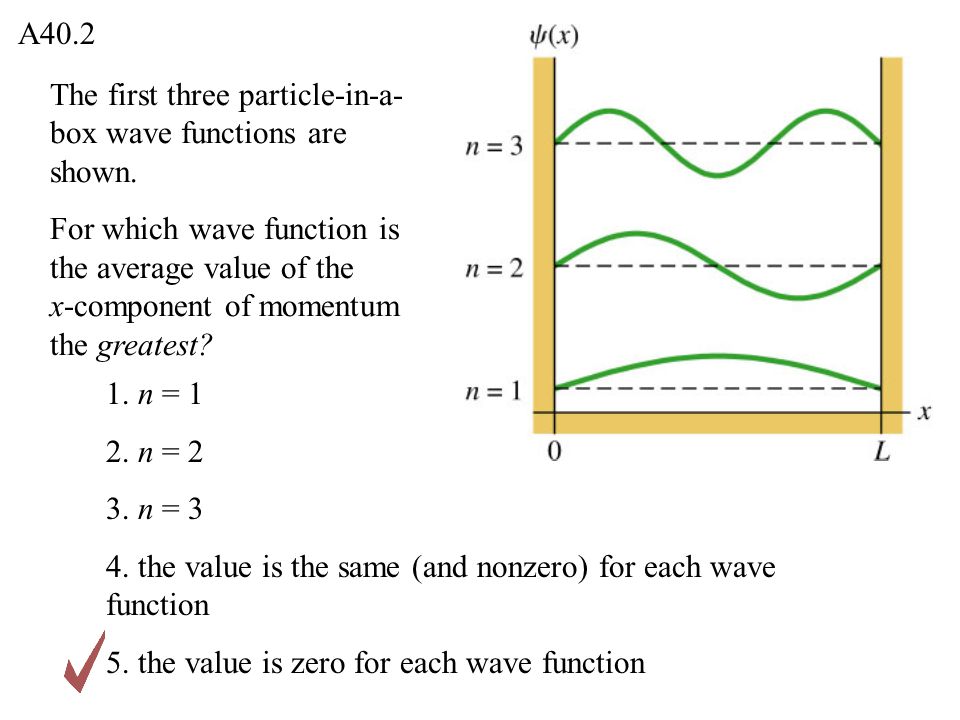
The first three particle-in-a- box wave functions are shown. For which wave function is the probability of finding the particle near x = L/2 the smallest? - ppt download
Consider Hydrogen ion in a box having one dimension, then find electron probability distribution? | Socratic
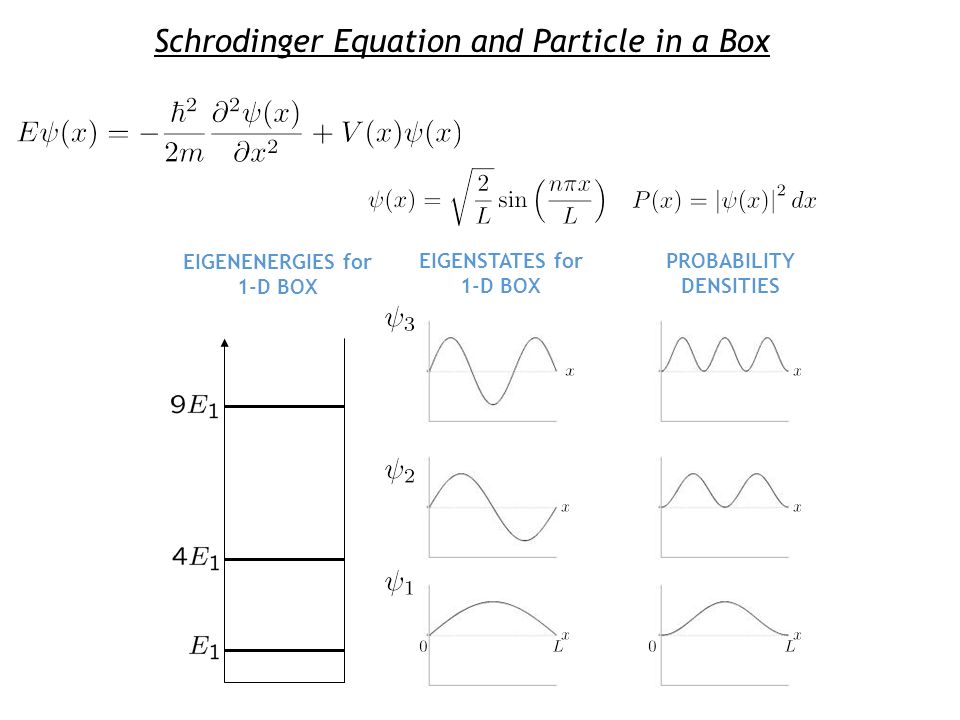
Reflection and Transmission at a Potential Step Outline - Review: Particle in a 1-D Box - Reflection and Transmission - Potential Step - Reflection from. - ppt download
Solved] Based on the particle-in-a-one-dimensional-box model, suggest where along the box the n=1 to n=2 electronic transition would most likely tak... | Course Hero
What is the probability of finding a particle in a one-dimensional box in energy level n=4 between x=L/4 and x=L/2? L is the length of the box. - Quora
![SOLVED:[3] On the following diagram for the probability density of a particle in a box; identify: (1) the pointswherewecannotfindthe particle, (2)thepointsweare mostlikelytofindthe particleand (3) write down the energy level n of the SOLVED:[3] On the following diagram for the probability density of a particle in a box; identify: (1) the pointswherewecannotfindthe particle, (2)thepointsweare mostlikelytofindthe particleand (3) write down the energy level n of the](https://cdn.numerade.com/ask_images/ff3fca7676af4612879843d82dfb5b7b.jpg)
SOLVED:[3] On the following diagram for the probability density of a particle in a box; identify: (1) the pointswherewecannotfindthe particle, (2)thepointsweare mostlikelytofindthe particleand (3) write down the energy level n of the