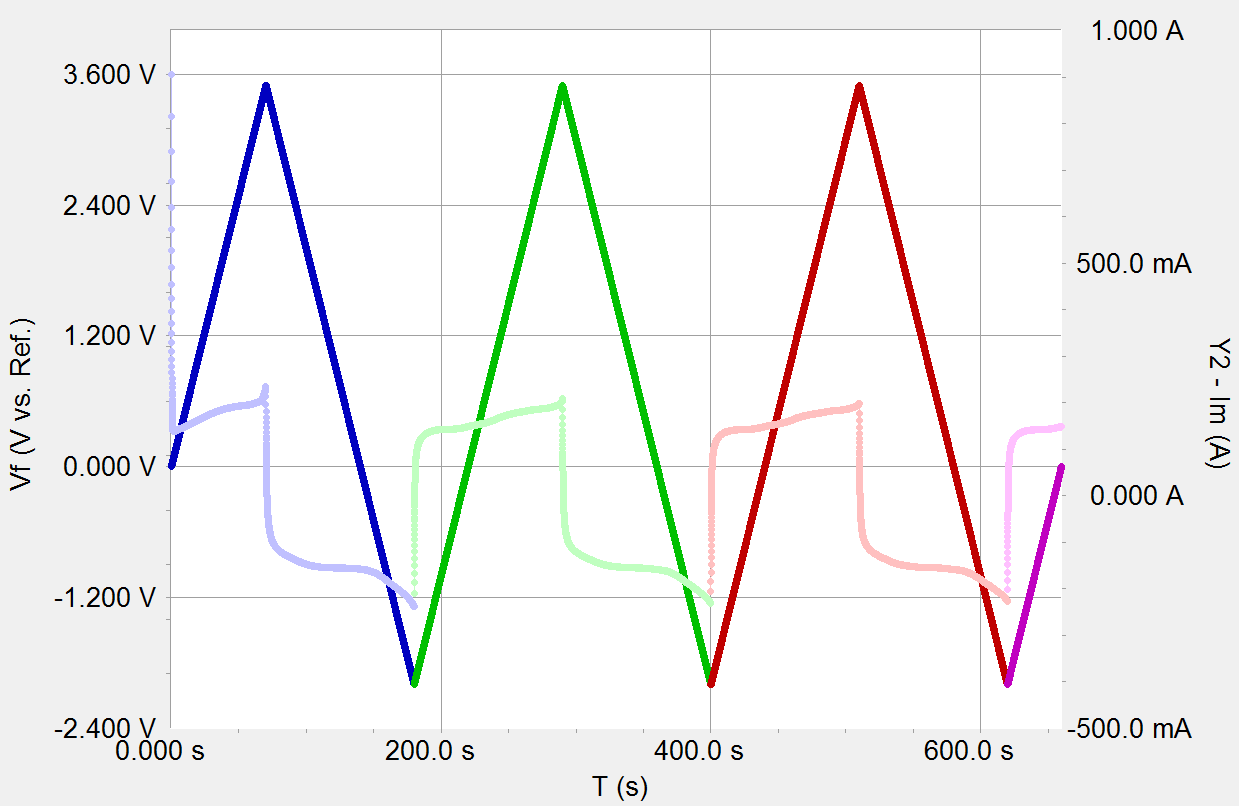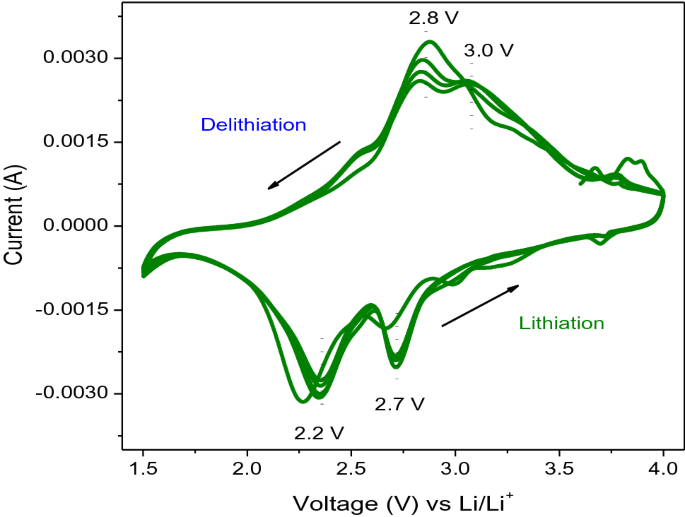
Inexpensive colloidal SnSb nanoalloys as efficient anode materials for lithium- and sodium-ion batteries - Journal of Materials Chemistry A (RSC Publishing) DOI:10.1039/C5TA10568D
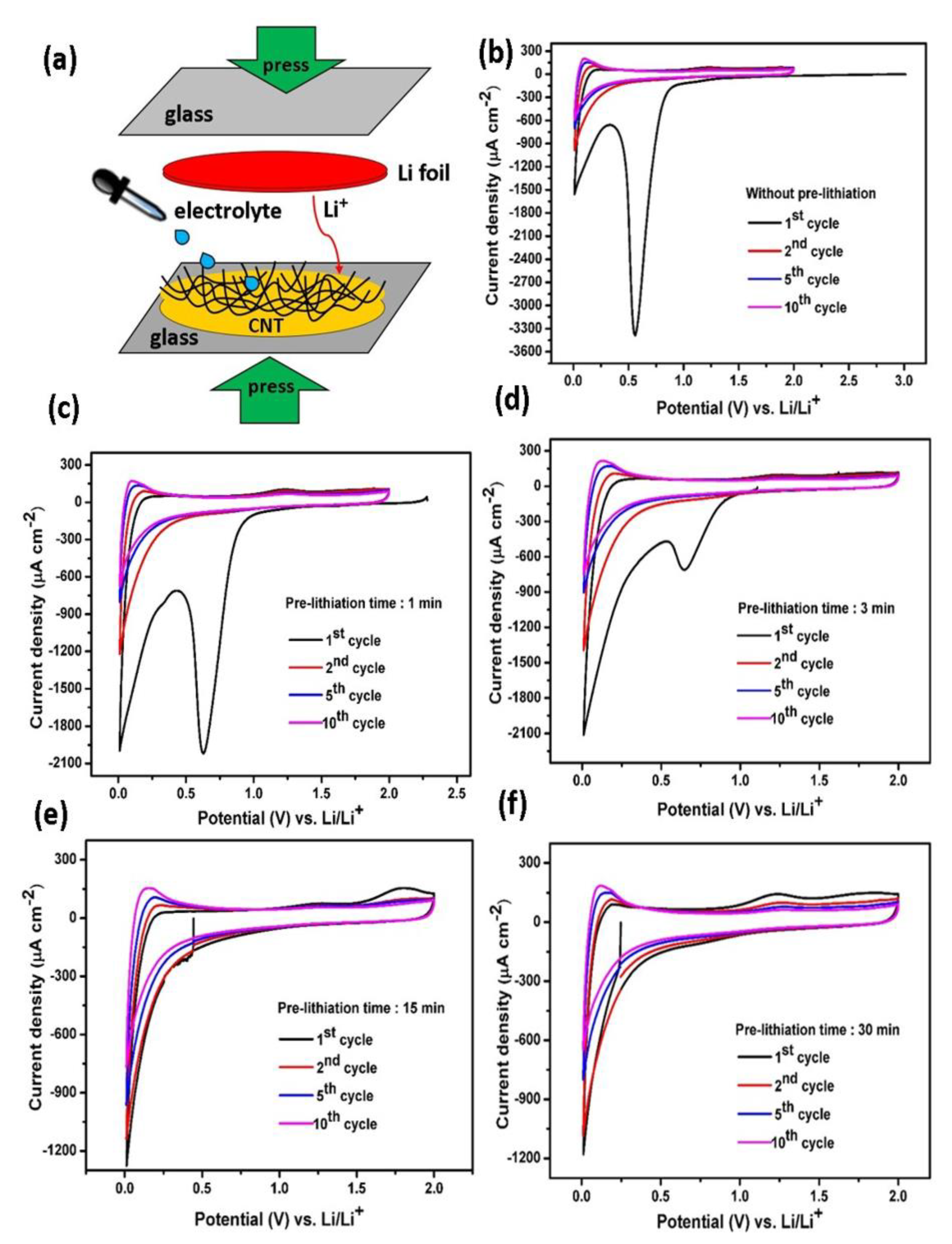
Polymers | Free Full-Text | Direct Pre-lithiation of Electropolymerized Carbon Nanotubes for Enhanced Cycling Performance of Flexible Li-Ion Micro- Batteries | HTML
Electrochemical performance of reduced silicon. (a) Cyclic voltammetry... | Download Scientific Diagram

Cyclic voltammetry curves of Li-S battery with a) NCP-CPE and b) liquid... | Download Scientific Diagram

Surface Chemical Analysis of Solid-Electrolyte Interphase Layer on Germanium Thin Films and the Effect of Vinylene Carbonate Electrolyte Additive | IntechOpen
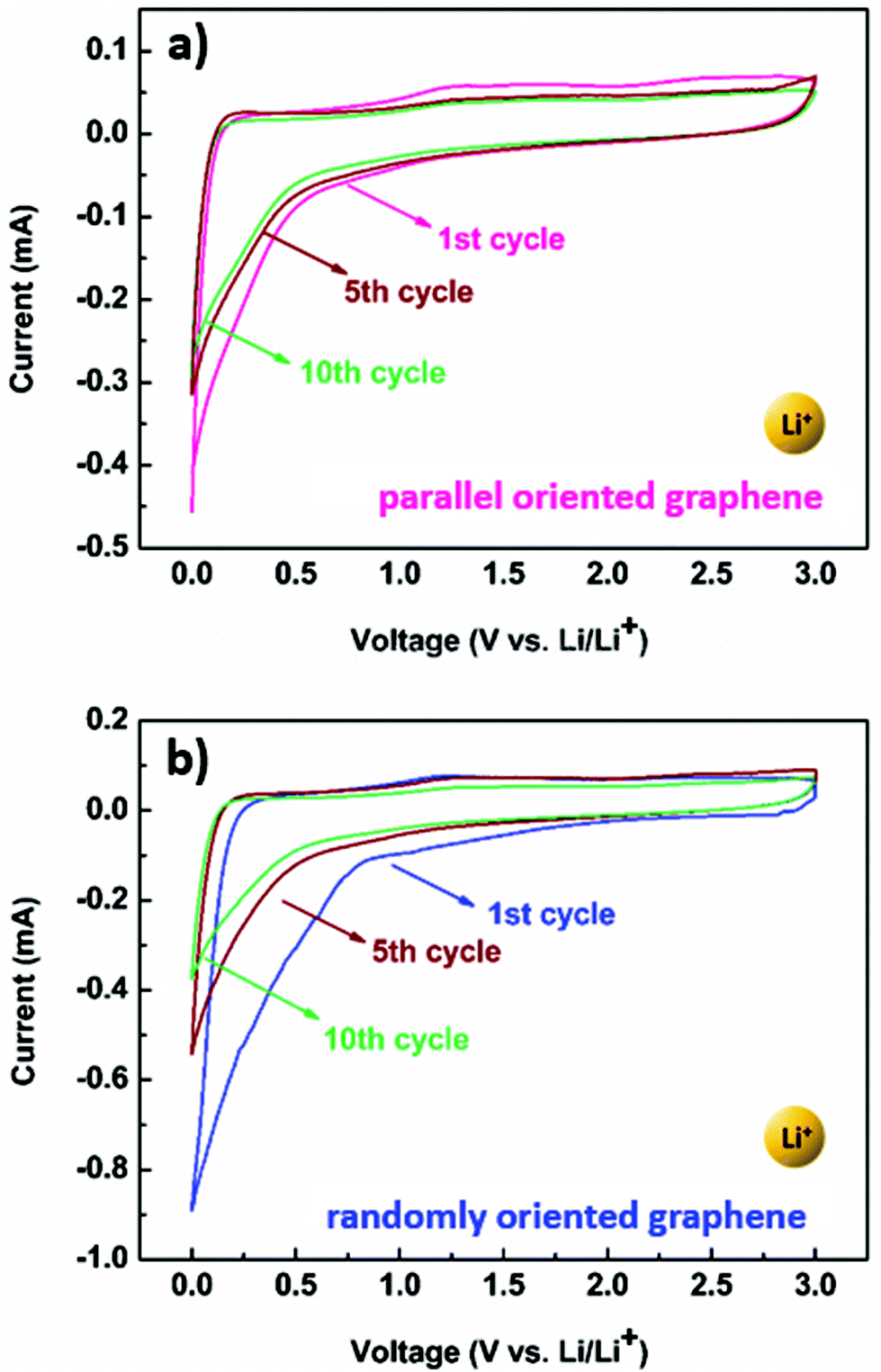
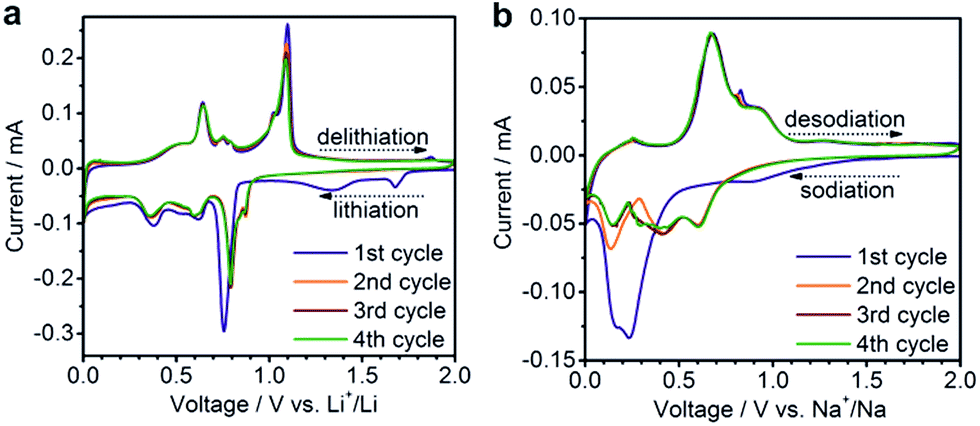
Comparison of reduction products from graphite oxide and graphene oxide for anode applications in lithium-ion batteries and sodium-ion batteries - Nanoscale (RSC Publishing) DOI:10.1039/C6NR07650E
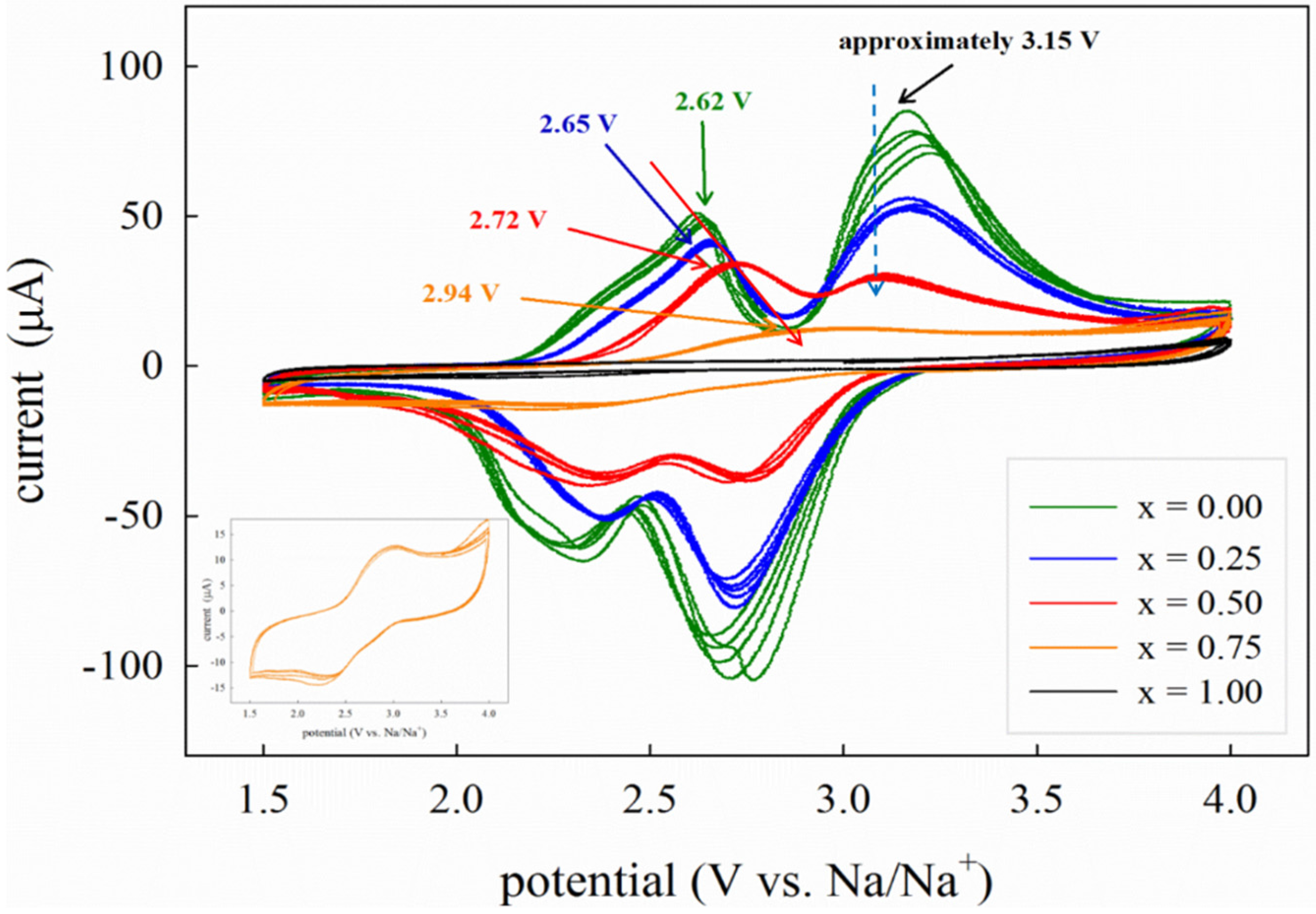
Batteries | Free Full-Text | Electrochemical Study of Na2Fe1−xMnxP2O7 (x = 0, 0.25, 0.5, 0.75, 1) as Cathode Material for Rechargeable Na-Ion Batteries | HTML
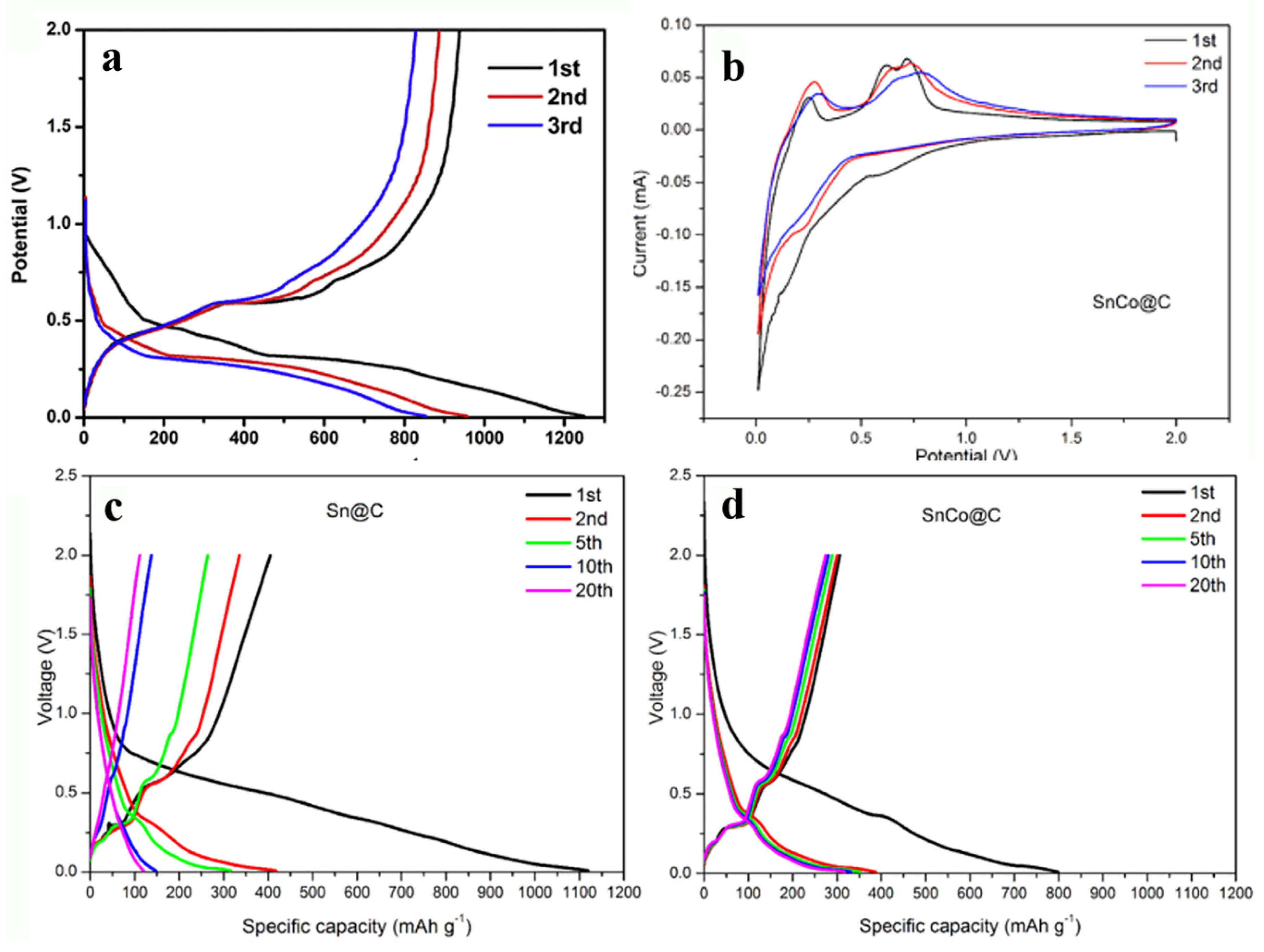
Applied Sciences | Free Full-Text | The Progress of Cobalt-Based Anode Materials for Lithium Ion Batteries and Sodium Ion Batteries | HTML

Figure 5 from Synthesis of Hierarchical Sisal-Like V2O5 with Exposed Stable {001} Facets as Long Life Cathode Materials for Advanced Lithium-Ion Batteries. | Semantic Scholar

Cyclic voltammetry of the FeMoO4 nanocubes at a scan rate of 0.1 mV s−1... | Download Scientific Diagram

Size-dependent cyclic voltammetry study of silicon microwire anodes for lithium ion batteries - ScienceDirect

Figure 5 from Reduced graphene oxide wrapped FeS nanocomposite for lithium-ion battery anode with improved performance. | Semantic Scholar
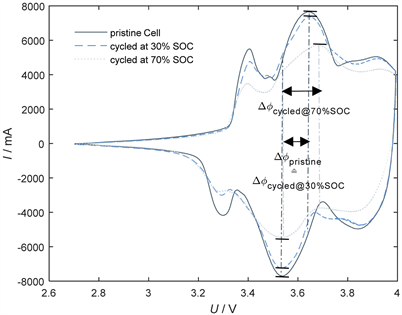
Ageing Behavior of LiNi0.80 Co0.15Al0.05O2 Cathode Based Lithium Ion Cells—Influence of Phase Transition Processes
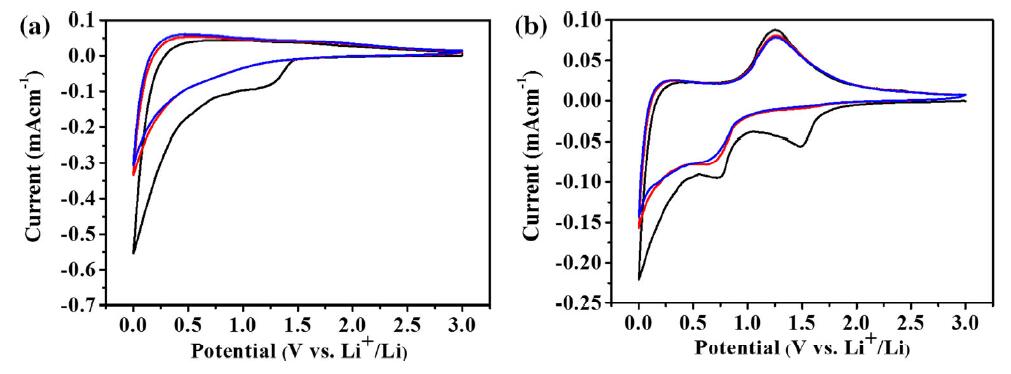
Scalable Synthesis of Hierarchical Antimony/Carbon Micro-/Nanohybrid Lithium /Sodium-Ion Battery Anodes Based on Dimethacrylate Monomer

Improving the Cycling Performance of Lithium-Ion Battery Si/Graphite Anodes Using a Soluble Polyimide Binder. - Abstract - Europe PMC

Forming solid electrolyte interphase <italic>in situ</italic> in an ionic conducting Li<sub>1.5</sub>Al<sub>0.5</sub>Ge<sub>1.5</sub>(PO<sub>4</sub>)<sub>3</sub>-polypropylene (PP) based separator for Li-ion batteries

Enhanced cyclic performance of SnO2-CuO-graphite nano-sheets as anode for Li -ion battery - ScienceDirect

Hybrid lithium-ion capacitors based on novel 1-butyl-3-methylimidazolium bis(nonafluorobutanesulfonyl imide) (BMImBNFSI) ionic liquid electrolytes: a detailed investigation of electrochemical and cycling behaviors - ScienceDirect